A rapid testing realization and COVID contextualization
Heading into 2020's final act, change may be on the horizon.
Good morning. It’s Thursday, September 24. Somehow, we’re a week away from the fourth quarter of 2020. Wow. Exciting things are beginning to happen as we approach Q4, and we’ll get to them soon. I guess the best thing to preach right now is patience.
Some weeks are slower, while others are packed with COVID happenings. I apologize again for not publishing last week, but it wasn’t due to a lack of content. I expect that stream of developments to continue in the coming weeks and months, which should be a positive thing. In Positivity Amid the Pandemic, the saying “no news is good news” doesn’t really apply.
I wish I could get to everything. But if there’s one thing that’s clear from this newsletter, it’s that I’m human. I can’t catch everything, but I’ve put together this week what I think is a good mix of science in theory and in practice. There’s plenty of testing news to go around, along with news about treatments in development and ones soon following, some further contextualization on the novel coronavirus and some quick hits.
This issue’s pictures are some of my favorite yet. We’ve experienced a great run of weather here in Evanston, and the shots show it. I’m a little biased, but Northwestern’s campus is unmatched (at least when it’s warm).



Trends
Party like it’s early June. Except don’t party, at least not inside or with a large group. The national case positivity rate, both an indicator of testing levels and community spread, at just over 4 percent, is its lowest since the week of June 7.

Hospitalizations are also the lowest they’ve been since mid-June, while deaths continue to fall and have a seven-day average of just over 700 daily fatalities, a marked decrease from when over 1,000 people were dying per day last month. Some of that can be attributed to hospitals having improved quite a bit at treating COVID patients.
This evidence speaks to the larger point that our COVID toolbox is expanding. I featured a Tweet a few weeks ago from Ashish Jha saying getting infected later on is better, and it’s so true. Death rates have improved considerably since March and April, and I have a feeling that even in November and December things may look different than they do now in late September.
With autumn officially here and cases expected to rise — how much is anyone’s guess — the need for frequent and rapid testing is paramount. Fortunately, it seems like more than just this newsletter, rapidtests.org and Michael Mina are on board at this point. That’s where we’re headed below.
The world realized what it wants is rapid testing
It’s coming. Just wait for it and then watch it happen. Even though it seems like we hear less of “the FDA wants x” or “company x says this could be the solution to our problems,” individual industries have decided rapid antigen testing is what they need to bring themselves back to life.
Before we get into those who are leading the recent charge, here’s a quick refresher on these antigen tests, which look for proteins of live virus as opposed to genetic material which can harmlessly linger in someone well past recovery. Here’s what they’re good for, and what they aren’t meant to be.
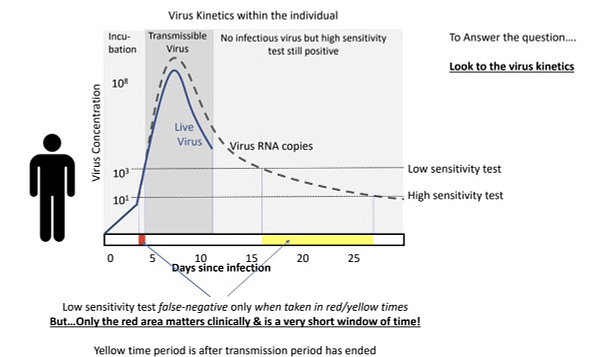
Look at this graph to understand the context when we say infection tests vs infectiousness tests. The small red time period (roughly 1 day) is when you’d get a false negative from the antigen tests as compared to gold standard PCR tests. Incubation periods vary, but the average person is unlikely to be infectious, or able to transmit virus, for the first five days of infection. PCR tests, with the highest of sensitivities, won’t pick up the virus likely the first four days, but they’ll find genetic material well after someone’s infectious period, which according to this display is typically five days to a week long. To reiterate, these numbers aren’t exact or the same for every infection, but the general concept is what matters.

Industries whose main source of revenue involves in-person activity have been leading the charge to procure rapid antigen tests as part of their safe reopening plans. I was ecstatic last week when the Big Ten announced it was bringing back fall football just a month after it postponed the season indefinitely. What caused university presidents’ minds to change? Daily rapid tests. Just listen to Northwestern team physician Jeff Mjaanes talk about the game-changing capabilities of these tools.
“Antigen testing detects certain proteins in the virus and can actually detect a level of virus that is thought to be the below the level of infectivity,” said Northwestern head team physician Dr. Jeff Mjaanes. “You’re basically catching a positive before it’s contagious. That’s a huge breakthrough. We can identify people before they are infectious, maintaining the health and sanctity of the team.”
So, I don’t know if I can TOTALLY agree with Mjaanes’ comment about the tests detecting people before they can transmit the virus. But I don’t know exactly what antigen tests they are using. Not all are created equal, and at least the lateral flow paper strip tests, the pregnancy test-like ones, currently don’t, to my knowledge, catch someone before they’re infectious. If you’re testing daily, though, you’ll catch most infected people before they have a chance to spread it to many others.
Boom, Big Ten football is back, and I get to cover Northwestern this season as a result of my favorite scientific development of 2020 thus far. I wrote for the NU sports blog I run about how the Big Ten may lead the way in America when it comes to regaining facets of life by using these tests. If this works, other sports can stage seasons, and then we’re talking about a host of other possibilities.
The other industry that has recently come out hard in favor of rapid tests is air travel. Some European airports have established impressive testing capacities of several thousand passengers per day, and some have even adopted rapid testing. With restrictive 14-day quarantines in several parts of the world devastating the travel space, leaders are looking to find ways to address that.
A rapid antigen test developed by Innova Medical Group could soon be used at London’s Heathrow Airport if it gains UK government approval. Furthermore, airlines are pushing hard for these tests to be a new standard at airports as a way to help bring international travel back. The International Air Travel Association (IATA) this week called for rapid, inexpensive tests for all passengers.
In the most concrete plan and step that I’ve come across, Germany-based Lufthansa plans to begin screening passengers with rapid antigen tests next month. Due to current supply constraints, first- and business class passengers will be tested (did we really think this wasn’t going to be the case?). It’s the first step in both helping re-establish consumer confidence in the pandemic age and charting the way forward as we adapt to live with the coronavirus.
And then how about theme parks? Or basically any in-person experience, whether it be theaters, stadiums, cruises, conventions, etc. Imagine them like the before times, when masks were meant for costumes and distancing was meant for avoiding that annoying friend you’ll meet only for lunch. How pleasant does that world sound.
Well, former Walt Disney imagineer Eddie Sotto is striving to bring that experience back, even before the pandemic is over. How? By forming bubbles using rapid testing as pre-entry screenings. Test negative? Go have fun like it’s 2019. Positive? Go home and we’ll see you next time.
“He envisions a minimally invasive procedure, such as providing a saliva sample or blowing into a Breathalyzer-like device, coupled with on-site equipment that could accurately diagnose coronavirus infection within a few minutes. And he wants to make it a fun experience.
The technology isn’t quite there yet, but it is advancing rapidly, and should be ready for prime time soon. Sotto says his goal is to be able to apply the strategies next season, possibly by spring.” - USA Today
Of course, the imagineer wants to make the screening process fun and creative, according to USA Today. Imagine blowing a bubble and the color it turns indicates a positive or negative result. Laugh now, but technology like this wouldn’t just be cute — it would literally bring back things people miss the most, and in a safe way.
Anyone and everyone is trying to develop their own innovate testing solutions to make things safe and restore public confidence. Who makes it how far, we don’t know. But let’s watch. I’ll happily take a rapid test before going to a sporting event or into a restaurant if it meant I didn’t have to worry about being in the presence of transmissible COVID.
Science closing in on COVID
Winter is coming, and so is science. Researchers are hard at work studying SARS-CoV-2 and everything about it, identifying its tendencies and its weaknesses. The Bloom Lab at the University of Washington last month published the first study providing evidence of immune protection from reinfection, and a couple weeks ago it studied the virus’ mutation patterns.
Without losing you (and me) in a super-detailed explanation, the paper looked at mutations in the receptor binding domain (RBD), which is what antibodies bind to and fight. Its findings included blueprints to developing mutation-proof antibody cocktails, drugs that work!


It also helps that SARS-CoV-2 is mutating very slowly, which bodes well for both drug and vaccine development as well as prospects of reinfection. There are some articles from large news organizations, like one yesterday from The Washington Post, which unfortunately miss the mark on mutation reporting. The piece will have some sensationalist headline about a mutation, when really the “new strain” is one that emerged in February and has been the predominant one in the United States. If anything it just confirms prior findings.


University of Pittsburgh scientists made headlines when they announced they found a biomolecule that is highly effective in treating and preventing SARS-CoV-2 infection in animals. There’s no guarantee those results translate to humans, as we well know, but it’s very encouraging. The antibody ingredient, known as Ab8, is 10 times smaller than a normal antibody and could be administered through routes besides an IV drip.
“Ab8 not only has potential as therapy for COVID-19, but it also could be used to keep people from getting SARS-CoV-2 infections,” said co-author John Mellors, chief of the Division of Infectious Diseases at Pitt and UPMC.
“At very low concentrations, Ab8 completely blocked the virus from entering cells. With those results in hand, a team at UNC tested Ab8 at varying concentrations in mice using a modified version of SARS-CoV-2. Even at the lowest dose, Ab8 decreased by 10-fold the amount of infectious virus in those mice compared to their untreated counterparts. Ab8 also was effective in treating and preventing SARS-CoV-2 infection in hamsters, as evaluated by scientists the University of Saskatchewan.”
A company backed by the school’s medical center has licensed the molecule for worldwide development, though it’s unclear how quickly the potential treatment could be ready. It appears human trials have yet to commence, but that could soon change.
Finally, the first late-stage data readouts from the big antibody cocktail candidates should soon come in. Eli Lilly, without releasing full data, announced its drug reduced levels of coronavirus and symptoms in participants who received the treatment as compared to those receiving the placebo. 1.7 percent of those who got the drug required hospitalization, compared with 6 percent of those who received a placebo — a 72 percent difference. The data is early and largely incomplete, but what we see so far is good. Regeneron could get a readout on some of its efficacy trials within the next couple of weeks.
Quick hits
One thing I believe has been lacking from pandemic coverage or common discussion is context. It can help provide nuance and shift us away from this all-or-nothing approach to the pandemic in which the only way to be responsible is not leave one’s property or thinking that mask-wearing is somehow a conspiracy.
But I think if there’s one topic that lurks in the minds of many is potential lingering affects of COVID-19, namely for the lungs and heart. The heart in particular has drawn a lot attention. What I’m about to say isn’t diminishing or downplaying the cardiac issues COVID may cause but comparing it to how we treat other illnesses. Think for a moment: what other illness do we pay this much attention to, and in real time?


If you don’t know Ed Yong, maybe you accidentally put the mask over your eyes. His pandemic coverage has been second to none if not also trailblazing. He wrote a piece all about COVID and the heart, and why much of our labeling of this virus is a result of an atypical amount of attention. I recommend you read it, as I think it provides great perspective on what we’re dealing with and how people react to it. Here are some of the quotes that stuck from me:
“These developments have only added to COVID-19’s mystique. News stories and scientific articles have spun a narrative about a bizarre virus that behaves like no other, and a supposedly respiratory illness that should perhaps be reconsidered as a vascular disease. But several cardiologists and virologists I’ve talked with say such claims are overblown. COVID-19 is a severe disease that should be taken seriously, but it’s not all that strange. It seems that way in part because it is new and extremely widespread, and so commands our full attention in the way that most viral illnesses don’t. Hundreds of researchers are studying it. Millions of people have been infected by it. And every study, every news story, and every unusual detail quickens the pulse.”
“Reassuringly, ‘there hasn’t been an obvious influx of patients being admitted to the hospital with unexplained myocarditis, despite the huge numbers who have had COVID-19,’ says Venkatesh Murthy, a cardiologist at the University of Michigan. “I don’t find it convincing that there is a major amount of serious clinically relevant myocarditis in people who are feeling well.”
“When millions of people become infected, rare events become commonplace, and phenomena that might typically have gone unnoticed suddenly become prominent. This creates a deceptive sense that the disease in question is stranger than most, and has uprooted the world because there’s something inherently odd about it.”
If you need proof that testing works, look no further than two colleges (among several others) that have managed to suppress COVID, even after early clusters of cases. The magic number at Cornell University and University of Illinois is two - that is, two tests per week for each student.
Read this thread from Carl below to learn how well Illinois is doing. After its “outbreak”, when the test positivity rate hit nearly three percent (lol), the seven-day positivity has consistently stayed under 0.5 percent. I’ve highlighted the school multiple times before as what was the best effort from any college at giving an on-campus a go. So far, good returns.

Finally, Johnson & Johnson began its Phase 3 vaccine trials yesterday. It will enroll 60,000 patients, the largest of any Phase 3 trial currently being run in the United States. The vaccine could be the first to require only one dose as opposed to two doses. Depending on the intensity of the US epidemic, J&J could know whether the vaccine is effective before the end of the year. I’d take a Christmas surprise from Santa Johnson.
Whew. Congrats if you made it all the way here. Enjoy the first weekend of fall. I’ll see you next week, when it’ll be October.
Once again, I appreciate your reading Positivity Amid the Pandemic. Have questions, comments and/or suggestions for me? Want to send me something to include in a future issue? Send me an email at EKarp@u.northwestern.edu or DM me on Twitter @karp_eli.
If you are enjoying Positivity Amid the Pandemic, share or subscribe!


